 The Born-Haber cycle allows us to understand and determine the lattice energies of ionic solids. As can be seen from the diagram, the differences in electronegativity between the elements of Groups 1-2 and those of Group 17 (as well as oxygen in Group 16) are sufficiently great. However, lattice energy cannot be directly measured. Chemists usually apply the term 'ionic solid' to binary compounds of the metallic elements of Groups 1-2 with one of the halogen elements or oxygen. Ionic solids tend to have high melting points and are rather hard. This Comment also clarifies how the charge of point defects fits with the standard. Ionic solids consist of positively and negatively charged ions held together by electrostatic forces the strength of the bonding is reflected in the lattice energy. These compounds have an additional stability due to the lattice energy of the solid structure. Classical experiments from solid-state electrochemistry can be used to determine the charge of ions in solids. RbI contains a metal from group 1 and a nonmetal from group 17, so it is an ionic solid containing Rb + and I − ions. The enthalpies of formation of the ionic molecules cannot alone account for this stability. The bonding within ionic or covalent solids may be stronger, but it is also directional, making these solids subject to fracture (brittle) when struck with a hammer, for example. Germanium lies in the p block just under Si, along the diagonal line of semi-metallic elements, which suggests that elemental Ge is likely to have the same structure as Si (the diamond structure). Metallic solids are known and valued for these qualities, which derive from the non-directional nature of the attractions between the kernel atoms and the electron fluid. Arrange the solids in order of increasing melting points based on your classification, beginning with molecular solids.Ī.Based on their positions, predict whether each solid is ionic, molecular, covalent, or metallic. Locate the component element(s) in the periodic table. Precipitation reactions occur when cations and anions in aqueous solution combine to form an insoluble ionic solid called a precipitate.Whether or not such a reaction occurs can be determined by using the solubility rules for common ionic solids.  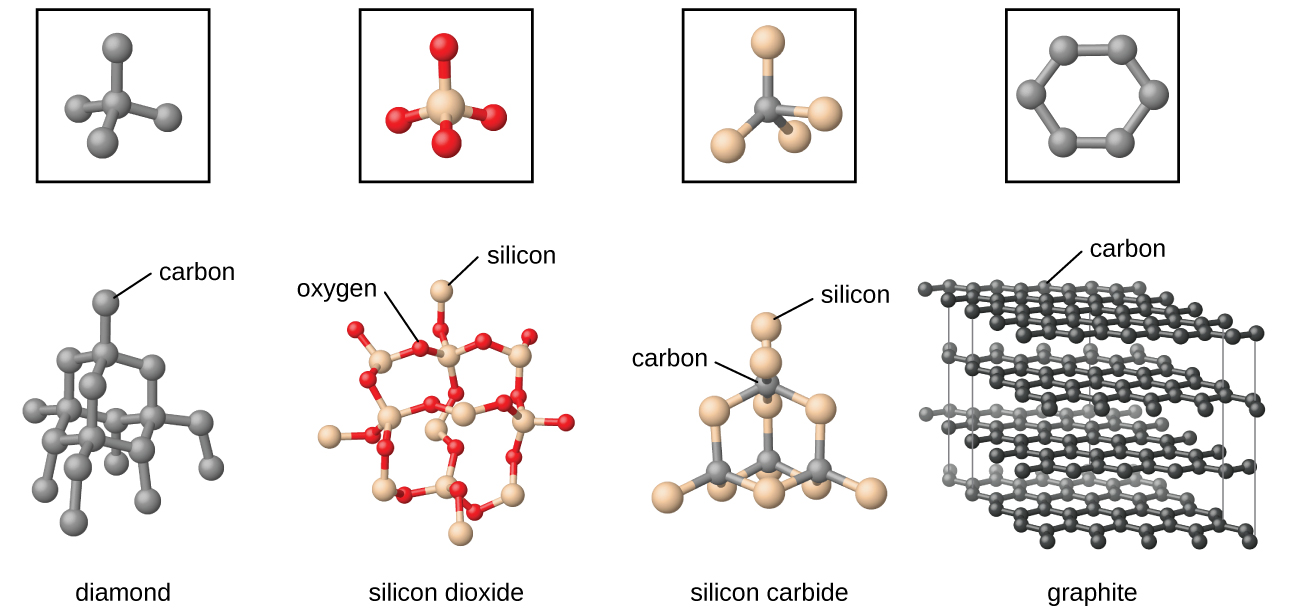
\) as ionic, molecular, covalent, or metallic solids and arrange them in order of increasing melting points.Īsked for: classification and order of melting points The following sections provide descriptions of the major types of crystalline solids: ionic, metallic, covalent network, and molecular. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
